
C. Scotti (Basel, CH)
Novartis Institutes for Biomedical Research, Translational MedicinePresenter Of 1 Presentation
10.2.11 - Anabolic Effect of LNA043: Results From an Imaging-Based Proof-of-Concept Trial in Patients with Focal Articular Cartilage Lesions
- B. Cole (Chicago, US)
- C. Scotti (Basel, CH)
- D. Laurent (Basel, CH)
- F. Saxer (Basel, CH)
- V. Juras (Vienna, AT)
- S. Trattnig (Wien, AT)
- S. Hacker (La Mesa, US)
- L. Pasa (Brno, CZ)
- R. Lehovec (Mladá Boleslav, CZ)
- P. Szomolanyi (Vienna, AT)
- E. Raithel (Erlangen, DE)
- J. Praestgaard (East Hanover, US)
- F. La Gamba (Basel, CH)
- J. L. Jiménez (Basel, CH)
- D. Ramos (Basel, CH)
- R. Roubenoff (Basel, CH)
- M. Schieker (Basel, CH)
Abstract
Purpose
LNA043 is a modified, recombinant human angiopoietin-like 3 (ANGPTL3) protein with chondro-anabolic effects on cartilage-resident cells and favourable safety profile. A first-in-human study demonstrated modulation of osteoarthritis-pathways, 7-Tesla magnetic resonance imaging (MRI) showed hyaline-like tissue-regenerates in a proof-of-mechanism study.
This study aimed at evaluating the chondro-regenerative capacity of intra-articular (i.a.) LNA043-injections in patients with cartilage lesions in the knee (NCT03275064).
Methods and Materials
This randomised, double-blind, placebo-controlled, proof-of-concept study treated 58 patients with a partial thickness lesion (43 [20mg LNA043]; 15 [placebo]), stratified by condylar vs. patellar lesion with 4, weekly i.a. injections. T2-relaxation-time (primary endpoint) was assessed as marker of collagen-network organization, lesion-volume was a secondary endpoint, both using 3-Tesla MRI. Assessments were performed at baseline, weeks 8, 16, 28 and 52. Lesion-volume was determined from manual image-segmentation, the cartilage volume of 21 joint-spanning sub-regions was measured from 3D isotropic MRIs with automated segmentation (prototype MR Chondral Health 2.1 [MRCH], Siemens Healthcare, Figure 1). An exploratory analysis evaluated the volumetric treatment-effect on the 3 weight-bearing medial femoral sub-regions (index-region).
Results
No difference in T2-relaxation-time was detected at any timepoint. Manual segmentation showed continuous femoral lesion-filling in LNA043-treated patients up to week 28 (p=0.08, vs placebo) without effects on patellar lesions. MRCH demonstrated cartilage-volume increase in the index-region (∆=123mm3 at week 28, N= 37, p= 0.05, Figure 2) without overgrowth in the lesion-free lateral condyles. The safety profile was favourable; eight drug-related treatment emergent adverse events were reported after LNA043, one after placebo, mostly joint swelling (9.3% vs 0), resolving spontaneously or with paracetamol/NSAID. No anti-drug antibodies were detected.

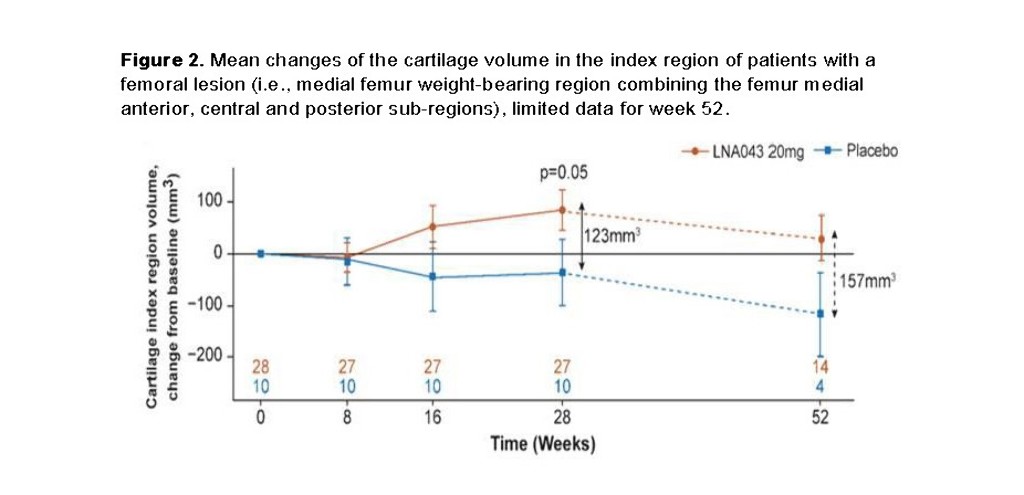
Conclusion
Treatment with 4, weekly i.a. injections of 20mg LNA043 resulted in regeneration of damaged cartilage in patients with femoral lesions. Automated assessment of cartilage volume in the index-region was more sensitive than manual segmentation. A Phase 2b study in patients with knee OA is ongoing.



