HYBRID CLOSED LOOP IMPROVES GLYCAEMIC CONTROL IN GASTROPARESIS IN TYPE 1 DIABETES
- Mark L. Evans, United Kingdom
- Mark L. Evans, United Kingdom
- Alison Jeffs, United Kingdom
- Makylah Lavender, United Kingdom
- Eleanor Gurnell, United Kingdom
- Helen Brown, United Kingdom
- Sarah Donald, United Kingdom
- Katy Davenport, United Kingdom
- Sara Hartnell, United Kingdom
Abstract
Background and Aims
Growing numbers of UK patients have been using the Minimed 670G Hybrid Closed Loop system since European commercialisation in 2018, with clinical "real world" experience extending beyond the initial data available from initial pivotal studies. Gastroparesis is a challenging complication which makes control of glycaemia difficult even with sensor augmented pump therapy. We report here our initial clinical experience with using the 670 in a subset of people with type 1 diabetes with advanced complications including gastroparesis.
Methods
We analysed data from 6 patients (all females already using insulin pumps, aged 47 + 4, diabetes duration 27 + 3 years, laboratory HbA1c 78 + 3 mmol/mol) started on 670G system as part of our clinical service from December 2018 onwards. All had both gastroparesis and multiple other complications of diabetes including laser-treated retinopathy, nephropathy, foot ulceration, faecal incontinence secondary to autonomic neuropathy, cerebrovascular accidents and angina. Gastroparesis had remained problematic in all despite optimised care (including intragastric botox injections and pyloroplasty in one individual and insertion of a gastric pacemaker in another). We compared data from sensor augmented pump therapy immediately prior to activating automode (closed loop) with subsequent data captured during automode.
Results
Time in range (3.9 to 10 mM) increased markedly from 35 + 3 % (consistent with challenging gastroparesis) to 64 + 4% at most recent downloads (average time using 670 system 7 + 1 month).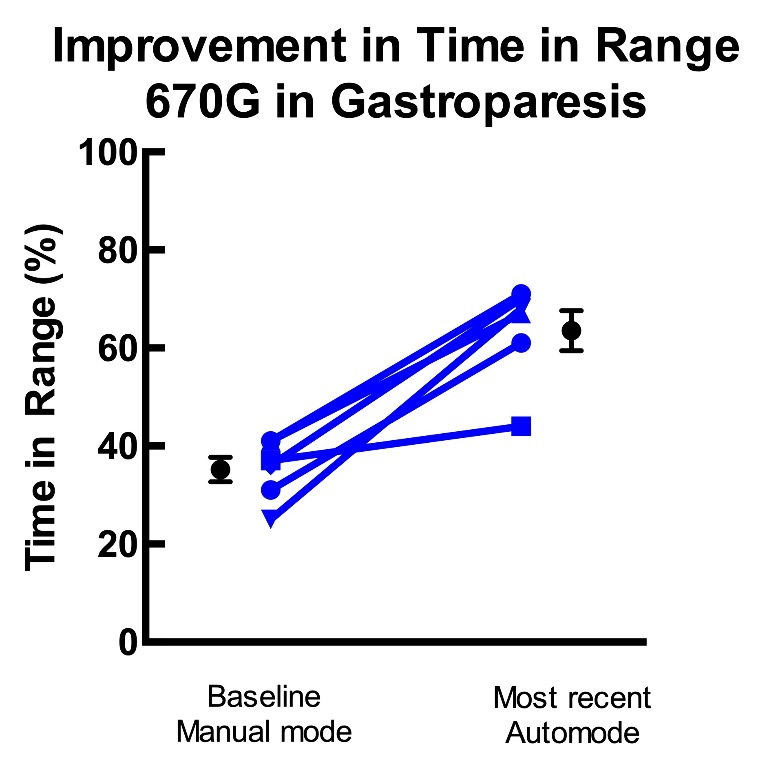
Conclusions
Hybrid closed loop insulin delivery may be extremely effective in improving glycaemic control even in those with gastroparesis and erratic glucose control.