NIGHT TIME GLYCEMIC CONTROL BEFORE AND AFTER MINIMED™ 670G SYSTEM USE BY CHILDREN WITH T1D 2-6 YEARS OF AGE
- John Shin, United States of America
- John Shin, United States of America
- Xiaoxiao Chen, United States of America
- Margaret Liu, United States of America
- Toni L. Cordero, United States of America
- Scott W. Lee, United States of America
- ROBERT A. Vigersky, United States of America
Abstract
Background and Aims
Adults who experienced increased overnight glucose variability or insulin delivery needs (akin to dawn phenomenon) during the MiniMed™ 670G system trial baseline run-in, exhibited improved overnight glycemic control after three months of Auto Mode use.1 The impact of Auto Mode use on the overnight glycemia of the youngest pediatric cohort of the MiniMed™ 670G system trials was investigated.
Methods
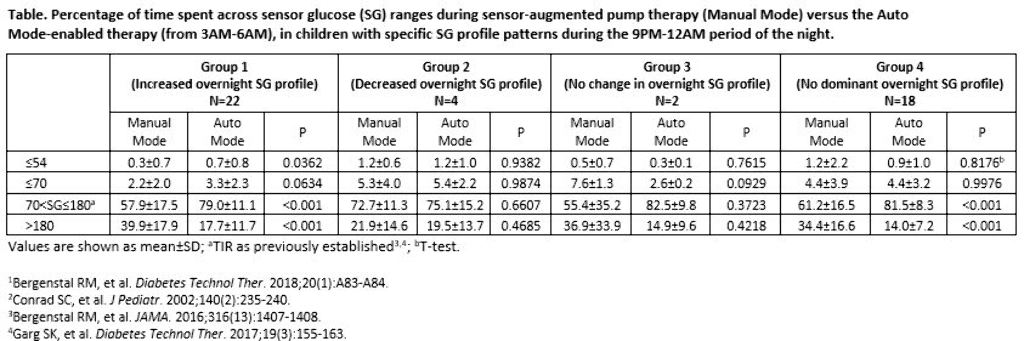
The 9PM-12AM sensor glucose (SG) profile of participants aged 2-6 years with T1D was analyzed, based on Conrad et al. 2002.2 Profiles were categorized as displaying a 1) >10 mg/dL increase in SG, 2) >10 mg/dL decrease in SG, or 3) No change in SG (≤10 mg/dL decrease or increase) and for >50% of the two-week baseline. If an SG profile did not present with an aforementioned pattern, it was categorized into a fourth group (No dominant SG profile). The percentage of early morning (3AM-6AM) time spent across SG levels were compared.
Results
There were 22, 4, 2, and 18 participants in Groups 1, 2, 3, and 4, respectively, during baseline. After three months of Auto Mode use, participants numbered 11, 1, 1, and 33, respectively. Significantly improved time in target glucose range (TIR, >70-180 mg/dL) was observed in Groups 1 and 4. Time spent at ≤70 mg/dL increased for Group 1, but was not significant.
Conclusions
Children with T1D displayed different profiles of overnight SG variability during the MiniMed™ 670G system trial. Regardless of the varied profile patterns observed, the MiniMed™ 670G system safely and effectively improved or did not change overnight TIR.