INTEGRATED TELEMEDICINE IN DIABETES CARE AND EDUCATION PROGRAM: WHAY DO CHILEAN FAMILIES LIVING IN REMOTE RURAL AREA THINK ABOUT IT?
Abstract
Background and Aims
Telemedicine (TM) is one more tool at modern medicine service at care of Type 1 Diabetes (DM1) moreover in remote areas. However, the reality from the families’ use and perceptions is not known
Methods
Qualitative exploratory study, from the content of TM conversation and interviews to 21 families that uses TM for care with DM1 between 2016 and 2018, from Aconcagua remote rural area.
Results
Our sample included children aged 11,4+- 4,04 years; treatment MDI (85,7%), CSII (14,3%); hospital visits 2,83 +- 1,38 per year. A1c average from 8,52 +- 2,17% (2016) to 7.9 +- 1,25% (2018). We analized 716 TM conversations mainly instant messaging (93,15%) with 12,35 ± 10.05 times per year and per family. The main themes for consultation were administrative (medical control and supplies) (35.3%), glycemic variability and insulin therapy (27.2%), urgencies management (20,5%), social support (11%) and education (5,8%). Satisfaction was optimal (100%) principally because of geographic and economic accessibility of TM, medical support availability to respond, educate and resolve daily difficulties, teamwork with professionals in the aim to improve metabolic control and quality of life. However, families identified more needs from TM to optimize its use, especially from remote rural areas.
Conclusions
Integrate TM into diabetes care program is a pillar of care and follow-up of youth and support families. However, there are still many pending challenges, including those recognized by the families, among the access, the new technologies, the technical conditions and the integration of peer support that should be reflected in future practices and studies.
CHALLENGES OF HYPOGLYCEMIA MANAGEMENT USING MOBILE APPLICATIONS
Abstract
Background and Aims
Many mobile applications (apps) for type 1 and 2 diabetes management incorporate results from BGMSs, however there is a paucity of evidence demonstrating benefits of apps for hypoglycemia management. Hypoglycemia management is associated with the performance of BGMSs in low blood glucose range (LBGR) and any app reliant on BG results is subsequently dependent. Previously we presented data demonstrating a reduced risk of hypoglycemia associated with the use of the CONTOUR®DIABETES app (CDA) whilst utilizing CONTOUR®NEXT ONE BGMS (CNO). Here we study the possible variation of hypoglycemia detection if BGMSs other than CNO were used with the app to obtain BG readings
Methods
All BGMSs were assessed for the likelihood of results ±15% of reference BG value of 50 mg/dl (CI 95%). We assumed that all assessed BGMSs were used with the app and all BG readings were analyzed identically as it has been performed in the CDA study using CNO.
Results
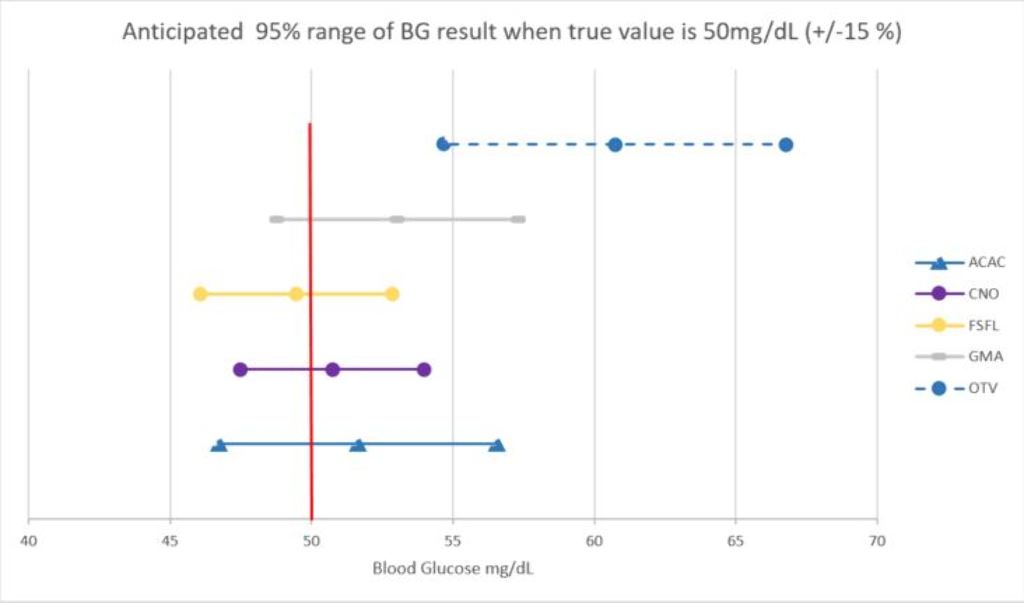
The probability of achieving ±15% of reference BG value of 50 mg/dl of some BGMSs were below 95% (Figure 1) and LL of BG readings of one BGMS (ACAC) were >50 mg/dl.
Conclusions
Not all BGMSs utilizing apps are capable of detecting hypoglycemia with a high probability thus not leading to potential reduced hypoglycemia occurrence as previously demonstrated. This analysis demonstrates that the selection of BGMS is critical in assessing the effectiveness of apps on glycemic control and particularly hypoglycemic management.
COACHING, CONNECTIVITY, CGM, AND CURATION: COMPLEMENTING CARE TEAMS IN THE U.S.
Abstract
Background and Aims
In response to the diabetes crisis and technological progress, recent years have brought about a new form of “connected diabetes care,” defined as digital diabetes management systems based around (1) smartphone apps, (2) devices with built-in connectivity, and (3) remote human and automated coaching and support. Given its potential to help improve health outcomes and the dearth of information about it to guide patients, providers, and payers, we describe and characterize connected diabetes care offerings.
Methods
Prominent connected diabetes care providers that have published results are listed and characterized. Similarities and differences are identified and the state of available evidence is evaluated.
Results
13 connected diabetes care offerings were analyzed for items including: health conditions managed, care team composition, connected medical devices, and evidence. We expect these companies will further expand offerings across chronic conditions, strive to integrate more deeply with the traditional healthcare system, deploy greater automation to promote scalability, and generate more robust evidence. Future trials should be randomized and have more standardized methodology in order to allow for stronger conclusions regarding comparative effectiveness.
Conclusions
The field of connected diabetes care has tremendous potential to improve outcomes, but it is in its infancy in terms of awareness, uptake, and effectiveness. However, existing evidence is sufficient to support further exploration and refinement of the model as the next step in team-based diabetes care. Our hope is that connected care can provide continuous clinical, lifestyle, and behavioral support for people with diabetes, delivering the right intervention at the right time.
MONTHLY, STRUCTURED VIDEO CONSULTATIONS FOR CHILDREN WITH TYPE 1 DIABETES HAVE A POSITIVE EFFECT ON METABOLIC CONTROL, DIABETES BURDEN AND TREATMENT SATISFACTION. RESULTS FROM THE VIDIKI STUDY, A MULTICENTER CONTROLLED TRIAL.
A DEEP LEARNING/RECURRENT NEURAL NETWORK METHOD TO PREDICT HOSPITAL ADMISSION FOR DIABETIC KETOACIDOSIS AMONG YOUTH WITH TYPE 1 DIABETES (T1D)
- Sarina Dass, United States of America
- Jon Bass, United States of America
- David Williams, United States of America
- Susana Patton, United States of America
- Ryan Mcdonough, United States of America
- Megan Schoelch, United States of America
- Mitchell Barnes, United States of America
- Sanjeev Mehta, United States of America
- Leonard D'avolio, United States of America
- Mark A. Clements, United States of America
Abstract
Background and Aims
We developed a deep learning model using a recurrent neural network to predict hospital admission for diabetic ketoacidosis (DKA) in the next 180 days among youth with T1D.
Methods
We selected 1523 youth 8-18 years old with new-onset (N=280) or existing (N=1248) T1D for model development/validation. All received care at a network of tertiary care diabetes clinics in the Midwest USA. Average admission rate for DKA was 6.33% (approx. 90) per 180-day window. We analyzed over 500 features/90-day period over 2 years, including demographics, clinical structured data (CPT codes, lab results and vital signs, medications, medical encounters), HbA1c trajectories, free-text clinical documents, and patient-reported outcomes from clinic intake forms. Text-based features were extracted using inverse document frequency on a bag-of-words for single words and bigrams, excluding stop words. The model produces a list of individuals who are rank-ordered by probability of DKA admission within 180 days.
Results
The cohort is characterized by age 13.72yr(IQR=11.30,15.78), 49% female, 80% white, 7% Hispanic, age at diagnosis 8.36yr(5.45,10.98), median A1c 69mmol/mol(60,82), and duration 4.58yr(2.08,7.67). Assuming 90 individuals admitted for DKA in 180 days, a rank-ordered list size=10 on an unseen test set yields precision (P)=100% and recall (R)=11%. For list size=25, P=48% and R=13%. ROC curve demonstrates AUC=0.77.
Conclusions
One can predict future DKA admissions among youth with T1D with high precision in a portion of the at-risk population. Whether specific behavioral or system-level interventions can reduce hospital admission rate remains to be determined.
ESTIMATION OF HEMOGLOBIN A1C FROM CONTINUOUS GLUCOSE MONITORING DATA IN INDIVIDUALS WITH TYPE 1 DIABETES: IS TIME IN RANGE ALL WE NEED?
Abstract
Background and Aims
Glycated hemoglobin (A1c) has long been considered the gold-standard metric for quality of glycemic control in diabetes and key predictor of long-term complications. At last-year ATTD, an international panel of experts convened to provide guidance on interpreting and reporting continuous glucose monitoring (CGM) data, recognized time in target range (TIR) as an important metric to complement A1c. In this work, we bridge the gap between the two metrics and present a method to derive estimates of A1c from TIR.
Methods
Data from the International Diabetes Closed-Loop Trial included CGM measurements from N=168 adults with type 1 diabetes over six months, and A1c samples at three and six months. Hemoglobin glycation and clearance were modeled through a first-order differential equation driven by a linear function of daily TIR. A population model was trained on half of the subjects and tested without modifications on the remaining half. The model was then personalized by identifying an individual glycation-rate coefficient using the first A1c sample. Measured and estimated A1c at six months were compared in terms of mean absolute difference (MAD), mean absolute relative difference (MARD), and correlation coefficient (R); 100 replicates of training/testing were performed to assess robustness.
Results
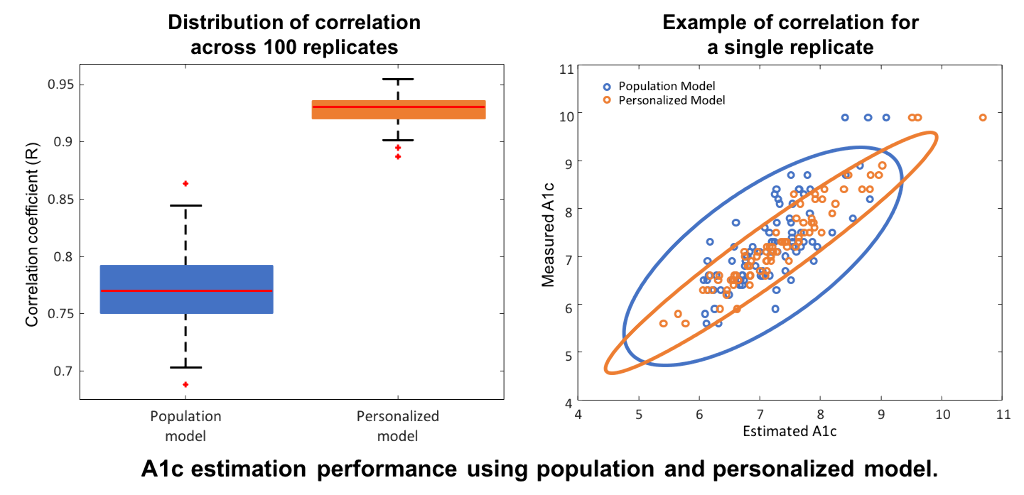
Compared to the population-based model, personalization with individual glycation rates decreased MAD (0.43% to 0.25%) and MARD (6.1% to 3.56%), and increased R (0.77 to 0.93 [figure]).
Conclusions
The TIR-A1c relationship is mediated by identifiable individual glycation coefficients. Following personalization, TIR provides high-precision A1c estimates, therefore representing a viable alternative to A1c in research and clinical-practice settings.
