PERFORMANCE OF THE LILLY AUTOMATED INSULIN DELIVERY (AID) SYSTEM: RESULTS OF EARLY PHASE FEASIBILITY STUDY
Abstract
Background and Aims
AID systems have led to demonstrated improvements in time in range without increasing hypoglycemia. Further advances in performance and usability will set the stage for more widespread adoption of this technology in diabetes self-management.
Methods
This inpatient early feasibility study evaluated the Lilly hybrid closed loop system comprising a high accuracy pump (developed by DEKA Research & Development Corp.), Lispro insulin, pump-embedded model predictive control algorithm, Dexcom continuous glucose monitor, and smartphone-based controller. Subjects underwent two similar 2-day inpatient protocols (10 adults with type 1 diabetes per protocol) including meal-related challenges (~30% over-bolus and 1-hour delayed bolus encompassing carbohydrate coverage and hyperglycemia correction) to simulate real-world diabetes self-management errors.
Results
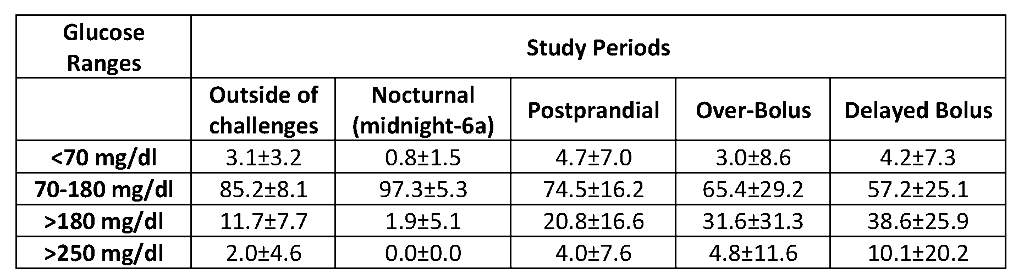
Subjects (25% male) had mean age: 44.7±14.2 years, T1D duration: 30.2±11.1 years, A1C: 7.2±0.8%, and insulin usage: 0.53±0.21 U/kg/day. The table below describes glucose control (percent time in different ranges [mean±SD]) while the subjects were on the AID system.
Conclusions
In this closely supervised feasibility study the Lilly AID system demonstrated expected algorithm performance in response to simulated diabetes self-management errors and promising glycemic outcomes. Clinical trial evidence in more ‘real world’ environments will provide additional information on the safety and effectiveness of the system.
PERFORMANCE OF OMNIPOD PERSONALIZED MODEL PREDICTIVE CONTROL ALGORITHM WITH MULTIPLE SETPOINTS AND MEAL AND EXERCISE CHALLENGES IN ADULTS AND ADOLESCENTS WITH TYPE 1 DIABETES
- Gregory P. Forlenza, United States of America
- Bruce A. Buckingham, United States of America
- Jennifer Sherr, United States of America
- R. Paul Wadwa, United States of America
- Alfonso Galderisi, United States of America
- Laya Ekhlaspour, United States of America
- Cari Berget, United States of America
- Liana Hsu, United States of America
- Melinda Zgorski, United States of America
- Joon Bok Lee, United States of America
- Jason O’connor, United States of America
- Bonnie Dumais, United States of America
- Todd Vienneau, United States of America
- Lauren Huyett, United States of America
- Trang Ly, United States of America
Abstract
Background and Aims
In preparation for future home studies, the safety and performance of the Omnipod hybrid closed-loop (HCL) personalized model predictive control (MPC) algorithm were assessed at multiple glucose setpoints and with meal and exercise challenges in adults and adolescents with type 1 diabetes (T1D) using an investigational device.
Methods
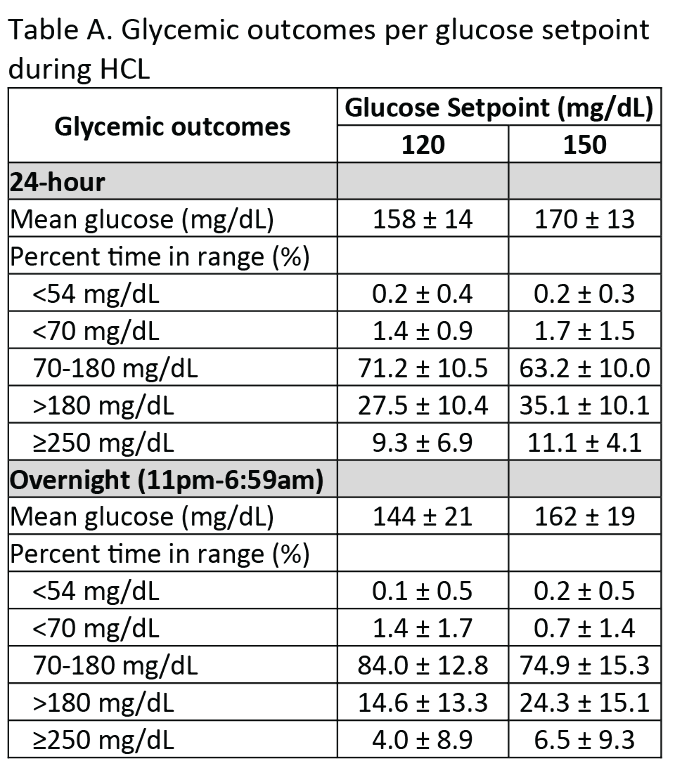
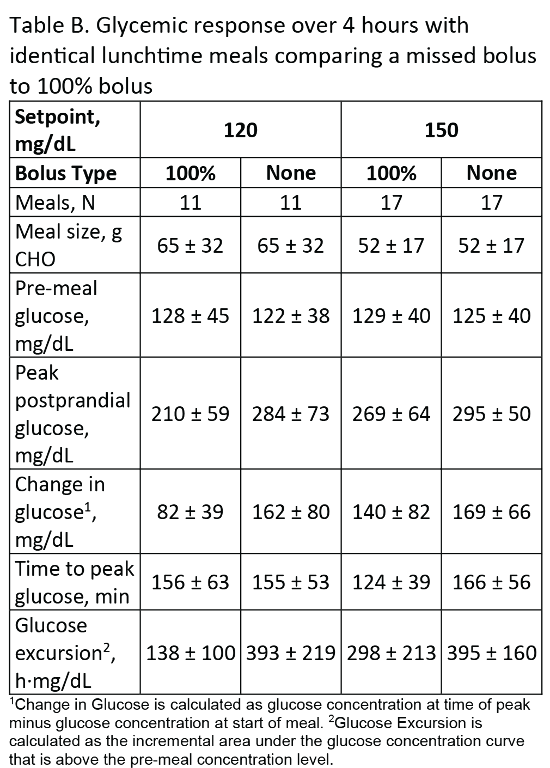
A 96-h HCL study was conducted in a supervised free-living hotel setting. Participants aged 12-85y with T1D and A1C<10.0% using CSII or MDI were eligible. At HCL start, the glucose setpoint was 150mg/dL, and was lowered to 120mg/dL after 48h. The system was stress-tested with 2 missed lunch boluses, high fat dinners, and daily moderate-intensity exercise. Endpoints were mean glucose and percentage time <54, <70, 70-180, >180, and ≥250mg/dL at each glucose setpoint. The 4-h glycemic response was assessed comparing a missed or 100% bolus for identical meals.
Results
Participants (n=20) were (mean±SD): age 28.5±15.0, T1D duration 16.7±12.2y, and A1C 7.4±1.0%. Glycemic outcomes at each glucose setpoint and following the missed meal bolus challenge are reported in Tables A and B, respectively. Despite 2 missed meal bolus challenges (20-136g carbohydrate), percentage time from 70-180 mg/dL was 71.2±10.5% with a setpoint of 120mg/dL, and 63.2±10.0% with a setpoint of 150mg/dL. Mean glucose was 158±14mg/dL and 170±13mg/dL at the 120mg/dL and 150mg/dL setpoints, respectively.
Conclusions
The Omnipod personalized MPC algorithm performed well and was safe in adults and adolescents with T1D when stress-tested at 2 different setpoints under challenging conditions. These data support exploration of lower system setpoints, as well as future home-based trials.
EFFICACY OF REAL-TIME MEAL DETECTION AND REMINDERS ON APPLE WATCH
Abstract
Background and Aims
Klue uses an Apple watch to detect hand motions indicative of eating or drinking. We hypothesized that real-time meal reminders delivered by Klue could decrease missed meal boluses and hemoglobin A1c (HbA1c) in adolescents and young adults with diabetes and 4 missed meal boluses in the previous 2 weeks.
Methods
This was a randomized, crossover, unmasked clinical study. Participants using continuous glucose monitor (CGM) with an insulin pump were randomized to either Klue for the first 6 weeks or standard care. There were 3 definitions for a meal occurrence: (1) Klue detection, (2) CGM rate of change of 2mg/dL/min for 20 minutes after cubic spline smoothing and (3) boluses for carbohydrates. Boluses were classified as premeal if ≤30 minutes prior to a CGM event, late if after CGM event but within 2 hours, and missed if no bolus within 2 hours.
Results
17 participants (mean age 17.7±4.6 years) were enrolled and 8 were randomized to start with Klue. The patients on Klue had a HbA1c decrease of 0.5% compared to the usual care arm (p=0.004). There were significantly fewer missed meal boluses (p < 0.00001) with Klue utilization. On average, Klue detected a missed meal bolus 18 minutes prior to a significant CGM rate of change and there was 1 Klue false-positive every 2-3 days.
Conclusions
Automated meal reminders improved HbA1c and reduced the number of late meal boluses. In addition to improving compliance, this technology has the potential to provide more rapid meal announcements to a closed-loop insulin delivery system.
UTILIZING BEHAVIORAL PROFILES IN A MULTISTAGE MPC TO IMPROVE POSTPRANDIAL GLUCOSE CONTROL
Abstract
Background and Aims
Postprandial glucose control is often difficult due to the time constants associated with insulin action. We aim to reduce postprandial hyperglycemia by creating an artificial pancreas (AP) framework that anticipates meals using patterns found from past eating behavior.
Methods
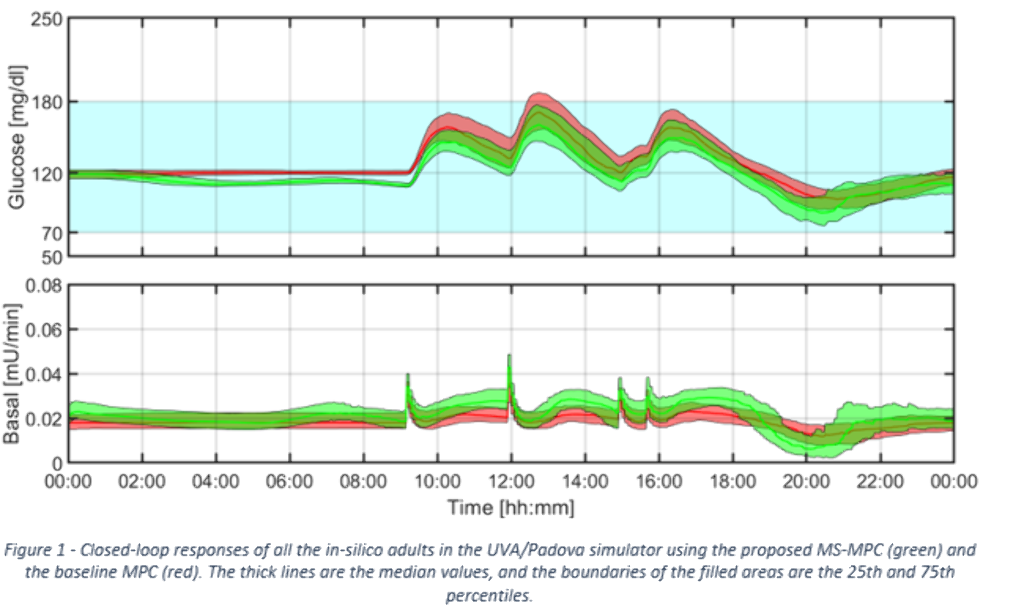
Oral glucose appearance profiles were created using daily meal records (75% training, 25% testing) from a subject that exhibits well-defined eating patterns (breakfast, lunch, and dinner). The novel AP is a hybrid multistage model predictive control (MS-MPC) that treats meal profiles as glucose subsystem disturbances. Simulations using testing data were performed for all 100 adult subjects in the UVA/Padova simulator. MS-MPC results were compared to a hybrid MPC not informed by meal profiles.
Results
Compared to the control MPC, the MS-MPC achieved lower 3-hour postprandial mean blood glucose (BG) (139.80(±12.53) v. 146.62(±12.76) mg/dL, p <0.01), more time in the 70-140 mg/dL range (60.09% (±17.08%) v. 48.82%(±18.31%), p<0.01), greater time in the 70-180 range (90.25% (±10.88%) v. 87.86%(±12.48%), p<0.01), and less time >180 (9.74% (±10.86%) v. 12.14%(±12.48%), p<0.01). The BG standard deviation for the MS-MPC group was higher than the control group (24.71 (±8.47) v. 23.51(±8.02) mg/dL, p<0.01). There was no significant difference between percent time spent <54, <70, and >250 mg/dL.
Conclusions
These preliminary, in-silico results suggest that the use of anticipatory meal profiles can increase tight time in range and reduce mean BG for some subjects that exhibit regular eating behavior in a MS-MPC in comparison to a standard, well-tuned MPC.
