- M. Elena Hernando, Spain
DEVELOPMENT OF AN EXERCISE ADVISOR SMARTPHONE APPLICATION FOR PEOPLE WITH DIABETES ON INSULIN THERAPY
- Michael C. Riddell, Canada
- Stephanie Edwards, United States of America
- Howard Wolpert, United States of America
- Michael C. Riddell, Canada
Abstract
Background and Aims
Regular exercise is an important part of healthy lifestyle for people with diabetes (PWD). However, the complexities around adjusting insulin doses and food intake to maintain glucose control during exercise are major barriers. Current paper-based guidelines are difficult to apply in practice and provide limited individualisation of recommendations, and there is an unmet need for software-based clinical decision support tools. Here we describe an application (app), which we have developed, to provide personalised proactive guidance with the goal of facilitating improved glucose control around exercise.
Methods
In the software development process, the decision tree from the current international consensus guidelines (Riddell et al.) was deconstructed and expanded from 8 branches in total to 3 separate decision trees for aerobic, mixed and anaerobic exercise with 20, 20 and 6 pathways, respectively. The app provides recommendations for insulin dose adjustments throughout the day and weight-based carbohydrate supplements, as well as pre-emptive bedtime guidance to prevent nocturnal hypoglycaemia following exercise. Guidance is customised based on insulin delivery modality (pump vs injection therapy), insulin-on-board status and includes specific exercise suggestions based on current glucose levels and hypoglycaemia risk. This latter functionality, specifically recommendations for mixed or anaerobic exercise activity to minimise carbohydrate supplementation requirements, could facilitate the use of exercise to support weight management goals in PWD on insulin.
Results
Not applicable
Conclusions
This personalised exercise advisor app addresses an important therapeutic need for a tool to facilitate exercise in PWD on insulin.
Reference:
Riddell et al. Lancet Diabetes Endocrinol. 2017;5:377-390.
INSULIN PUMP USAGE PATTERNS AMONG PERSONS WITH DIABETES (PWD) PREDICT DETERIORATING SENSOR-BASED GLUCOSE CONTROL 2-4 WEEKS FOLLOWING CLINIC VISITS: A MACHINE LEARNING MODEL
- Sarine Babikian, United States of America
- Sarine Babikian, United States of America
- Vikram Singh, United States of America
- Mark A. Clements, United States of America
Abstract
Background and Aims
We developed a machine learning algorithm that predicts risk for decreased sensor glucose time in range (TIR) following a clinic appointment, using primarily clinic-uploaded device data.
Methods
We selected 3175 PWD using pump+CGM who had 7079 clinic upload events of pump data to Glooko between 1/19/2018-6/28/2019. We trained a random forest-based algorithm to predict risk for a 10% decrease in TIR within any 14-day window in the next 30 days, relative to the average TIR 30 days pre-appointment.
We built features from insulin usage trends over the 30-90 days pre-appointment: means and standard deviations of values and counts for boluses (extended, manual, normal, without carbs, with override, with correction), basals (scheduled, suspend, temporary increase and decrease), and carbohydrate amounts. We also included average CGM active time, diabetes type, and gender.
Results
The training cohort had median age=24 years (IQR:12-43), 56.3% female, 80.9% type 1 diabetes (T1D), average blood glucose median=168.3mg/dL (IQR:147-191.2). We performed out-of-sample validation using 680 individuals (1516 clinic upload events). The validation cohort had median age=23 years (IQR:11-42), 55.5% female, 78.9% T1D, average blood glucose median=168.6mg/dL (IQR:147.4-190.7). The algorithm predicted individuals at risk of decreased TIR with a precision of 0.76 and a recall of 0.64. This correlates to sensitivity=64%, specificity=75%, and positive predictive value=76%.
Conclusions
The present model indicates that it is feasible to predict future deterioration in glycemic control at clinic visits with limited data; additional datapoints may improve predictions. Predicting worsening glycemic control via machine learning may help clinicians identify their most at-risk patients for more intensive intervention.
TRENDS OF COMPLICATIONS OF DIABETES IN TYPE 1 SUBJECTS WITH DIABETIC FOOT ON INSULIN PUMP THERAPY: A 5 YEARS REGISTRY ANALYSIS
- Alessandro Scorsone, Italy
- Alessandro Scorsone, Italy
- LUCIA Spano, Italy
- PERNA Leonardo, Italy
- VITO Aiello, Italy
- MANUELA Gambino, Italy
- GABRIELLA Saura, Italy
- MATTIA Fleres, Italy
- Anna Di noto, Italy
- FRANCESCA Provenzano, Italy
- Davide Brancato, Italy
- VINCENZO Provenzano, Italy
Abstract
Background and Aims
The onset of a diabetic foot ulcer is a severe complication.Our aim was to study if diabetes complications are better managed in Type 1 diabetes subjects on insulin pump therapy presenting with a diabetic foot problem.
Methods
We extracted data on T1 DM, from our electronic registry during the last 5 yrs.AIM: to study if insulin pump users had different mangement of diabetic foot.
Results
Population: 193 insulin pump users admitted to our outpatient diabetic foot clinic (23.3% first access). Mean age was 37.7 ± 6,9 (M ± SD). 90% of users ( 73% not users) underwent a diabetic foot screening procedure before, 70% vs 63% screened for diabetic retinopathy (p < 0.05). Mean HbA1c was not different (7.9 ± 1,6 vs 8.0 ± 1.7 %) . LDL was < 100 mg/dl in 55% of both. Among T1 DM subjects (users vs not users) BMI was > 30 in 13,3% vs 18,2%, eGFR < 60 ml/min 66,7% vs 33 %, CV event in 3,3% vs 4,5%, Microalbuminuria 26,7% vs 31,3 %, diabetic retinopathy rate and blood pressure levels were not different. No maior amputation rate was registered in users 3% in non users, lesser degree amputation in 3,3% and 3.5%.
Conclusions
Insulin pump users may have a better management of diabetes related complications, apart from glucose control, due to a more intensive follow-up scheduled for them that does not overlook any action of diabetes care. This may improve the outcome of diabetic foot and diabetes-oriented databases may help us in controlling health care procedures.
A WEB APPLICATION FOR THE IDENTIFICATION OF BLOOD GLUCOSE PATTERNS THROUGH CONTINUOUS GLUCOSE MONITORING AND DECISION TREES
- Ignacio Hidalgo, Spain
- Ignacio Hidalgo, Spain
- Marta Botella-serrano, Spain
- Francisco Lozano-serrano, Spain
- Esther Maqueda, Spain
- Juan Lanchares, Spain
- Remedios Martínez-rodríguez, Spain
- Arantza Aramendi, Spain
- Oscar Garnica, Spain
Abstract
Background and Aims
This increased demand for CGM devices means an opportunity for data and computer scientists, who can contribute to the development of decision-making support systems based on the data collected from the devices. Our aim is to applied Machine Learning techniques to this data and find patterns that leads to advisory systems.
Methods
Using both the entered data and the blood glucose values collected by the device automatically, the application presented here uses decision trees to detect the patterns and entails a starting point in the creation of ensemble models with more predictive power, also based on decision trees. Furthermore, the methodology makes a segmentation of the data set in blocks, determined by the different meals done throughout the day, adding more information to the set of variables used to train the models.
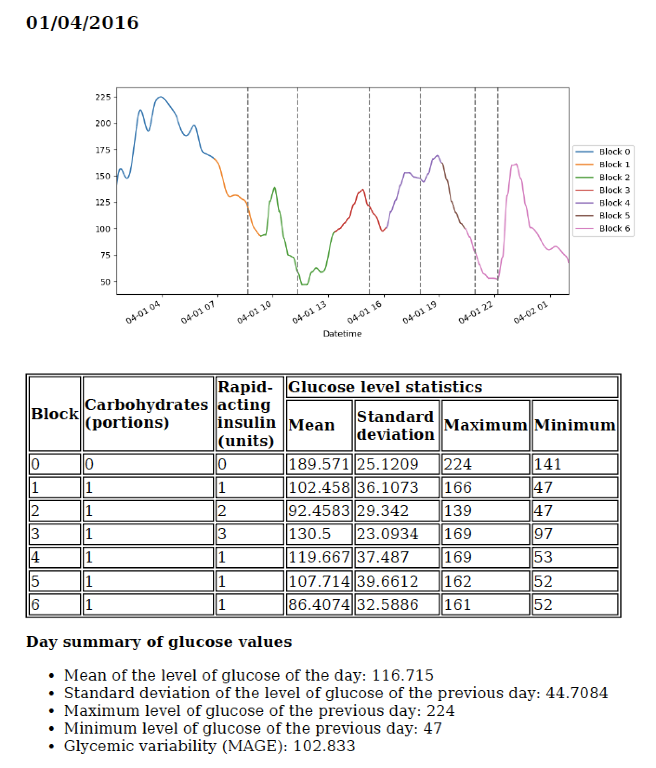
Results
The application developed in this project generates a report of the patient’s glucose patterns and provides a web application that allows the user to upload the data obtained from his device and download the report on his computer or smartphone.
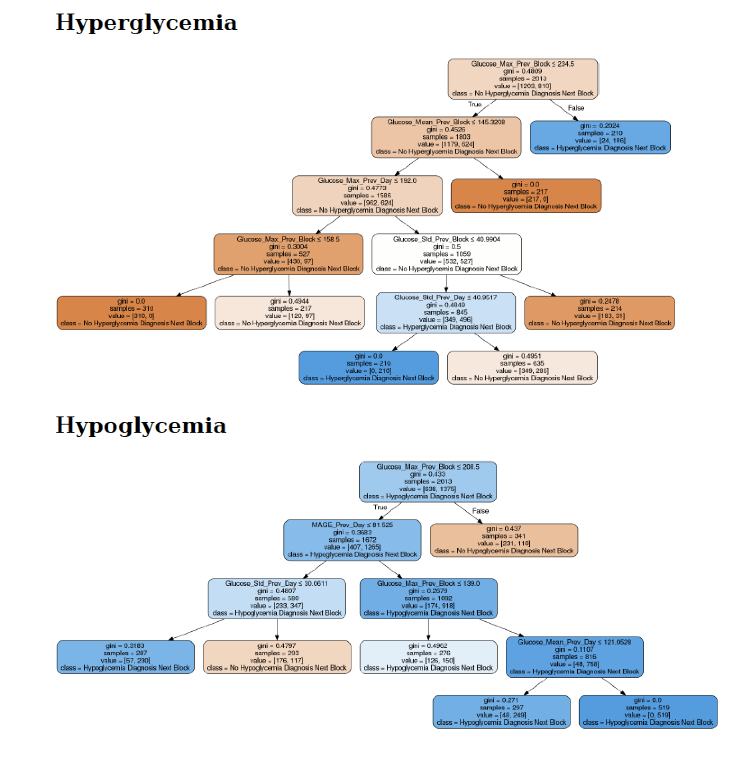
Conclusions
As a result, the application can discover repetitive patterns in the daily life of the patient, which can help him to take early preventive measures for risk situations in a period close to the next meal.
Thanks: RTI2018-095180-B-I00 and Fundación Eugenio Rodriguez Pascual